
Yu-Chen Wang
Farm Advisor Plant Pathology
Yu-Chen is the Plant Pathology Advisor serving Monterey, Santa Cruz, and San Benito counties. Her educational background includes a M.S. in Agriculture with a focus in Plant Protection from California Polytechnic State University, SLO and a M.S. in Horticultural Crop Science from National Taiwan University. She previously worked for Nematology lab at UC Kearney ag center and that sparked her an interest in soilborne pathogens. At Cal Poly SLO, her research focused on strawberry disease management for Macrophomina root rot and Botrytis Fruit rot. In her role as Plant Pathology Advisor, she implements an applied research and extension program in plant pathology to develop and extend practical, science-based knowledge to the agricultural community in the tri-county areas. Her program provides diagnostic service for California leafy greens producers and conducts research on the identification and management of vegetable and strawberry diseases.
Research update on organic fungicide evaluation for management of celery late blight

Research update on organic fungicide evaluation for management of celery late blight
Yu-Chen Wang, Renée L. Eriksen, Chris Greer, and Alex Putman
Celery late blight can cause significant problems during the fall and spring production period in the Salinas and Santa Maria Valleys. The disease is caused by the fungus, Septoria apiicola. Early disease symptoms include small, irregularly shaped, chlorotic spots on the leaves and petioles. Older leaves and stalks are usually infected first. As the disease progresses, the lesions enlarge, later merge to blight large areas of tissue, leading to plant death (Photo 1). A characteristic feature of these lesions is the small, black round structures in the center (Photo 2). These structures are the reproductive bodies of the fungus, and the size and color are similar to black pepper.


Management. The primary source of the pathogen is contaminated celery seeds and undecomposed celery debris. Cultural controls include using pathogen-free seeds and rotating away from celery for more than 1 year. The disease is favored by cool temperatures and prolonged wetness from rain, dew, fog, and sprinkler irrigation. Dense crop canopies reduce air movement and can also increase disease development by prolonging leaf wetness periods. Fungal spores are spread by splashing water from rainfall or overhead sprinklers. Movement of equipment or workers can also spread the spores from diseased to healthy plants.
There are a number of fungicides that have activity against the disease. Apply protectant fungicides, especially if sprinkler irrigation is used or rain has occurred or is forecasted. Managing this disease in organic production is even more challenging as fungicide options are limited. The following section addresses research needs for organic management of celery late blight by evaluating the field efficacy of some organic fungicides.
Research update on organic fungicide evaluation.
A fungicide trial was conducted at the USDA-ARS Spence Road research farm to evaluate the efficacy of select organic fungicides for managing celery late blight in the summer of 2025. Celery ‘Sonora’ was transplanted on May 1. Eleven organic fungicide treatments, a standard conventional rotation treatment, and an untreated control (UTC) were arranged in a randomized complete block design with four replications (Table 1). Each 20 ft-long plot consisted of two lines of celery on a narrow bed (40-inch centers). Plant spacing was 7 inches. A buffer bed on either side of the plot was left untreated to ensure there was no drift between treatments. Septoria inoculum (1.4 x 105 spores/mL) was prepared by soaking and stirring dried infected celery leaves for 30 min in water then straining through two layers of cheesecloth. Inoculum was applied with a hand-pump backpack sprayer to all treatments on July 3 using a single hollow cone nozzle at 15 gal/A followed by light moistening the plant canopy. Fungicide treatments were applied with a CO2-pressurized backpack sprayer calibrated to deliver 35 gpa at 30 psi using TeeJet 8004E flat fan nozzles on a two-nozzle boom. Fungicide applications were made on June 10, June 26, July 10, and July 24. Initial celery late blight severity was evaluated on July 28 on 10 plants from the middle of each treatment plot based on a 0-5 rating scale (0=no symptoms, 1=1%-10% leaf blight, 2=10%-20% leaf blight, 3=20%-30% leaf blight, 4=30%-50% leaf blight, 5=petiole blight). The severity evaluation was made on an outer leaf, a middle leaf, and an inner leaf on each plant. Disease severity was expressed as the average rating of the 10 plants. Data were third root transformed, analyzed using analysis of variance (ANOVA), and the Tukey HSD test to separate means at P<0.05.
Results. Late blight was slow to develop after inoculation, but with the conducive environmental conditions created by overhead irrigation twice a week and adequate dew periods for sporulation and infection, disease severity was low to moderate near harvest maturity. On the July 28 rating date (88 days after planting), the average disease severity of the nontreated control was 1.7 (Fig. 1). There were significant differences in the average disease severity among treatments. All treatments reduced celery late blight disease severity numerically, but Kocide 3000 and the conventional fungicide rotation treatment had significantly lower celery late blight severity than the nontreated control. The conventional standard treatment had the lowest late blight severity and was therefore the most effective treatment against disease symptoms. Phytotoxicity was not observed with any treatment in this trial.
Table 1. Treatment list
| Product and rate/A | Active ingredient | OMRI listed | Application timingz |
| Untreated control | - | - | - |
Conventional standard Tilt (4 floz/acre) Bravo Weather Stick (3 pt/acre) | Propiconazole Chlorothalonil | No | AC BD |
| Badge X2 (3.57 lb/acre) | Copper oxychloride | Yes | ABCD |
| Badge X2 (3.57 lb/acre) | Copper oxychloride | Yes | AB |
| Badge X2 (3.57 lb/acre) | Copper oxychloride | Yes | CD |
| Cueva (2 gal/acre) | Copper octanoate | Yes | ABCD |
| Microthiol (6 lb/acre) | Sulfur | Yes | ABCD |
| Experimental: S-3 | Lipopeptide | - | ABCD |
| Experimental: S-8 | Lipopeptide | - | ABCD |
| Kocide 3000 (1.5 lb/acre) | Copper hydroxide | Yes | ABCD |
| Oso 5% (13 floz/acre) | Polyoxin D zinc salt | Yes | ABCD |
| Regalia (3 pt/acre) | Reynoutria sachalinensis plant extract | Yes | ABCD |
| Double Nickel (6 qt/acre) | Bacillus amyloliquefaciens | Yes | ABCD |
zApplication timing: A=June 10, B=June 26, C=July 10, and D=July 24

2022 Lettuce Variety Tolerance to Pythium Wilt
Richard Smith1, J.P. Dundore Arias2, Kelley Richardson3, Yu-Chen Wang1 and Tricia Love4
1 - Farm Advisor UCCE Monterey, 2 - Plant Pathology Professor CSUMB, 3 - Research Geneticist USDA ARS, Salinas and 4 - Staff Research Assistant, UCCE Monterey
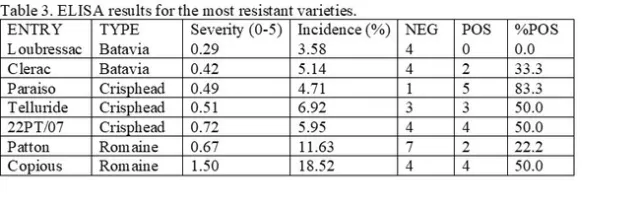
Summary: A lettuce variety trial was conducted at the USDA Spence research station on a site that had significant infection with Lettuce Pythium wilt (Pythium uncinulatum) in 2021. Visual evaluations of lettuce plants that were wilting or dead showed that a number of head lettuce varieties had significantly tolerance to Pythium wilt: 22PT/07, Paraiso, Telluride, Molera and 22PT/08. Romaine varieties were generally more sensitive to Pythium wilt than head lettuce varieties, but some varieties that held up to the disease included Copious, Patton, 1024 Momentus and SR2-21-33B. Two green leaf types also held up well to Pythium wilt: Clerac and Loubressac. INSV incidence (percent symptomatic plants) was 89% correlated with Pythium incidence. The most resistant Pythium varieties were also the most INSV resistant. While INSV severity and incidence in crisphead type varieties were significantly lower than in romaine varieties, on average, significantly more crisphead samples tested positive in laboratory tests for the virus than romaine samples. Laboratory evaluations of infected plants confirmed the dominant soilborne disease at the site was Pythium wilt. Pythium wilt was also isolated from plants that had no obvious visual symptoms in the above ground portion of the plant. No Fusarium wilt was detected in any plants sampled. In separate evaluations on commercial farms, levels of Pythium wilt increased to extremely high levels by the end of the production season in the Salinas Valley in 2022. Even varieties that were shown to be highly tolerant in the Spence variety trial showed significant susceptibility under severe late-season conditions.
Methods: Lettuce Variety Trial: A lettuce variety trial evaluating tolerance to Pythium wilt was conducted at the USDA Spence Research Station on a site that had significant infection with this disease in 2021. The trial was planted on July 12, 2022 using a modified planter on a two-bed sled with Stanhay planter shoes; seed was fed to the shoes by dropping seed by hand into a funnel and hose that carried the seed down to the shoes. Seed was pre-weighed out in coin envelopes and arranged in boxes to follow the plot map for the plot. Seed was received from 10 seed companies and a total of 53 varieties were included (Table 1). Each plot was one forty-inch bed wide (with two seedlines) by 25 feet long. There were four replications of each variety arranged in a randomized complete block design. After planting, thirty gallons of 7-7-0-7 was applied as an anticrustant and 3 pints of Kerb wase applied to control weeds. The field was sprinkler irrigated until August 9th when drip tape was installed and was used for the remainder of the crop cycle to irrigate the field. Two fertigations of 120 lbs of N/A as UN 32 were applied to the field.
Total plants in each plot were counted on August 9. Visual evaluations of Pythium wilt were conducted on three dates: August 30 and September 6 and 13 by counting the number of plants that exhibited wilting per plot which was converted to mean percent infected plants per plot by dividing by the total plants. Laboratory confirmation of Pythium wilt was conducted by collecting wilting plants from one rep of the plot on August 30 and September 6, and plants from all plots (regardless of visible wilting symptoms) on Sept 13. These plants were evaluated by J.P. Dundore-Arias Plant Pathology lab at CSU, Monterey Bay. On September 13, selected plants that were wilting, but showed no signs of obvious Pythium root rot and were therefore suspected of having a vascular wilt were sent to Alex Putnam who evaluated them for Fusarium wilt. Also on September 13, four plants were sent to the TriCal Diagnostic lab for general wilt evaluation. The plots were also evaluated for INSV by Kelley Richardson of the USDA. Twenty plants per plot were flagged in three replications (60 plants total). Each was rated for INSV symptom severity (0-5, 0=no symptoms, 5=dead) on September 7 and 14. INSV incidence was calculated per plot as the percent of plants showing symptoms (severity of 2 or more). On September 14, leaf tissue was collected from three plants per plot per replication of the most resistant lines (lowest severity) to test in the USDA lab for the presence of INSV using ELISA.
On-Farm Evaluations: Three on-farm varieties trials conducted by seed companies were evaluated as described above. The focus of the evaluation was to observe the interaction of INSV and Pythium tolerant/resistant varieties under conditions of high disease pressure. The dominant disease at the three sites was Pythium wilt which was confirmed by visual symptoms of rot on the roots. The plants were also evaluated for the presence of Sclerotinia and vascular wilts.
Table 1. Varieties included in the trial.
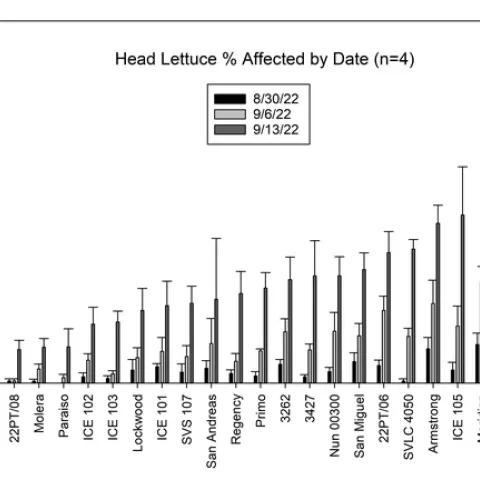
Results: Lettuce Variety Trial, Visual evaluations: showed significant differences among the varieties of head, romaine, and leaf type lettuces (Figures 1, 2 and 3). Head lettuce varieties differed in susceptibility, but also on the timing of infections. For instance, a number of head lettuce varieties had low levels of infection on August 30 but by September 13 exhibited a high diseases incidence with a large number of infected plants (Figure 1). 22PT/07, Paraiso, Telluride, Molera and 22PT/08 all had ≤10% on the last evaluation date (63 days after first water). Romaine lettuce were more sensitive to Pythium wilt and only Copious, Patton and 1024 had levels of Pythium wilt ≤10%, while Momentus and SR2-21-33B had levels ≤20% (Figure 2). Two green leaf lettuce types maintained levels of infection ≤10%, Clerac and Loubressac. Laboratory evaluations:Pythium wilt was consistently isolated from infected plants collected on the three evaluation dates. The number of Pythium-infected plants increased over the weeks and by the last evaluation, the pathogen was isolated from nearly all plants, regardless of having mild to severe foliar wilting or root rot symptoms. On September 13, selected wilting plants that did not have obvious symptoms of Pythium wilt were sent to Alex Putnam's lab, but of the 13 plants evaluated none had Fusarium wilt. One plant sent to the TriCal Diagnostic lab for general wilt evaluation was confirmed for Verticillium; plants with symptoms associated with a vascular wilt were not common in this trial.
There were significant differences amongst varieties for INSV severity and incidence at both rating dates (<0.0001). On average, batavia and butterhead lines had the lowest INSV severity and incidence (Figure 4). On average, crisphead varieties had significantly lower severity (1.76) and incidence (32.23%) than romaine types (3.12, 66.03%). This matches previous years' trends that crisphead varieties, on average, are more resistant than romaine. Cos, green leaf, and red leaf varieties had high severity and near 100% incidence. It is important to note that the two red leaf varieties tested had red and green leaves and were not completely red. This might explain why this data does not match previous trends of red leaf varieties having lower severity than green. Of the crisphead varieties tested, Paraiso, 22PT/07, and Telluride had the lowest INSV severity and incidence below 10% (Figure 5). 102, Molera, and Lockwood had low INSV severity and incidence below 20%. Patton and Copious were the only romaine varieties with low INSV severity and incidence below 20% (Figure 6). No romaine types had incidence below 10%. 1024, SR2-21-33B, and Momentous had INSV severity below 2 and incidence below 50%. Of all varieties tested, the Batavia varieties Loubressac and Clerac had the lowest INSV severity and incidence below 5% (Figure 7). ELISA was run on leaf tissue from the most resistant lines to check for INSV. Only Loubressac had all negative samples (Table 3). Surprisingly, romaine varieties, on average, had fewer positive samples than crisphead varieties. Paraiso, which had one of the lowest INSV severities and incidence below 5% had 83.3% positive samples.
The September 13 Pythium and September 14 INSV datasets were analyzed together to look for similar trends. Data sets were 89% correlated. When separated by type, batavia and butterhead varieties on average had the lowest INSV and Pythium incidences and crisphead varieties had significantly lower INSV and Pythium incidences than romaine varieties (Figure 8). The most INSV resistant varieties were also the most Pythium resistant (Figure 9).
On-Farm Evaluations: INSV (Rijk Zwaan - RZ) and Pythium (Momentus, Copius and Patton) tolerant/resistant varieties were evaluated in trials in commercial fields at the end of the growing season in the Salinas Valley during the period of most intense disease pressure. The varieties were compared with a susceptible variety. At Ranch 1 all varieties had greatly reduced wilted/dead symptoms compared to the field variety (Table 2). The Rijk Zwaan varieties had much reduced INSV symptoms as well. Ranch 2 had substantial wilted/dead plants but less INSV than Ranch 1; the Rijk Zwaan varieties had wilted/dead plants that ranged from 14.5% to 20.4% compared to 78.2% for the field variety. At Ranch 3 the field variety had 68.7% of wilted/dead plants and 15.7% with INSV only symptoms. Patton and Momentus had 42.9 and 49.3%, wilted/dead plants, respectively and did not have fewer INSV infected plants than the field variety. The Rijk Zwaan varieties ranged from 32.0 to 37.6% wilted/dead plants but had much fewer INSV only infected plants. The INSV symptoms on the Rijk Zwaan varieties showed very slight INSV symptoms (few spots on outer leaves) but were confirmed with Agdia strip assays. Plants at Ranch 3 were also evaluated for Fusarium and Verticillium wilts, but none were positive. The soil was also evaluated, and no Fusarium wilt of lettuce was detected and Verticillium levels ranged from 2.1 to 2.5 ms/gram of soil, which is not sufficient to cause disease on lettuce.

Figure 1. Visual evaluation of Pythium wilt: Mean percent infected plants of head lettuce per plot on three dates.
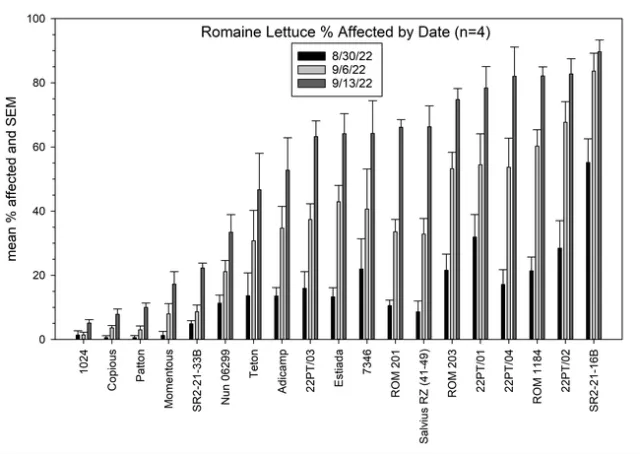
Figure 2. Visual evaluation of Pythium wilt: Mean percent infected plants of romaine lettuce per plot on three dates.
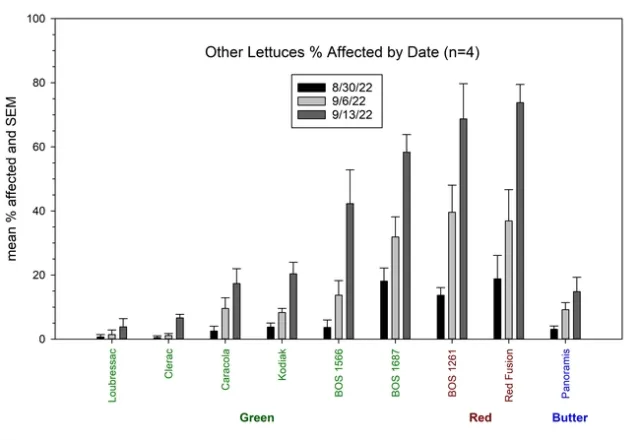
Figure 3. Visual evaluation of Pythium wilt: Mean percent infected plants of leaf lettuce per plot on three dates.
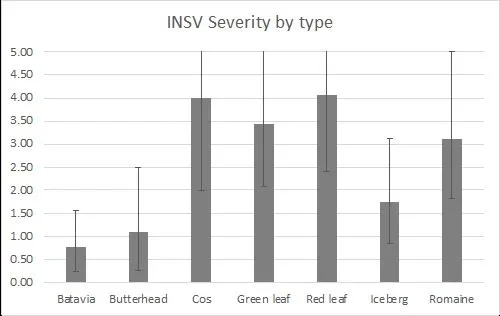
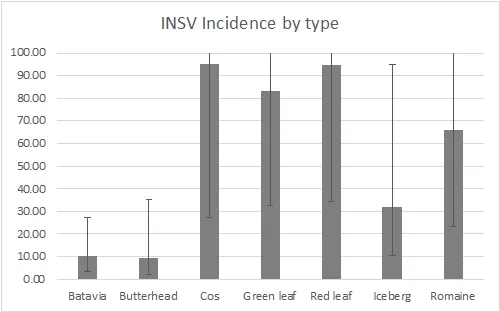
Figure 4. INSV severity (0-5 scale) and incidence (0-100%) of all lines separated by lettuce type. Final rating on September 14. (Error bars represent 95% confidence intervals)
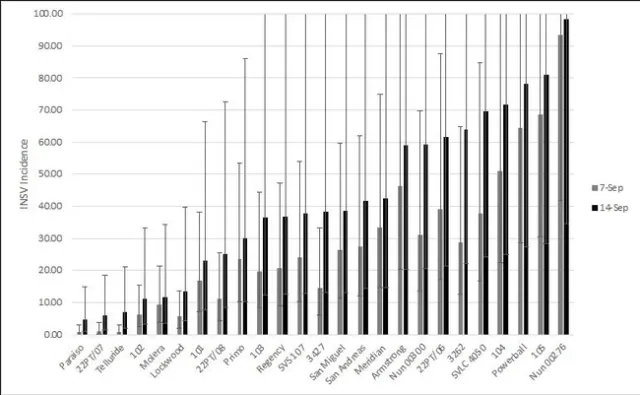
Figure 5. Mean INSV incidence (percent infected plants) of crisphead lettuce on two rating dates. (Error bars represent 95% confidence intervals)
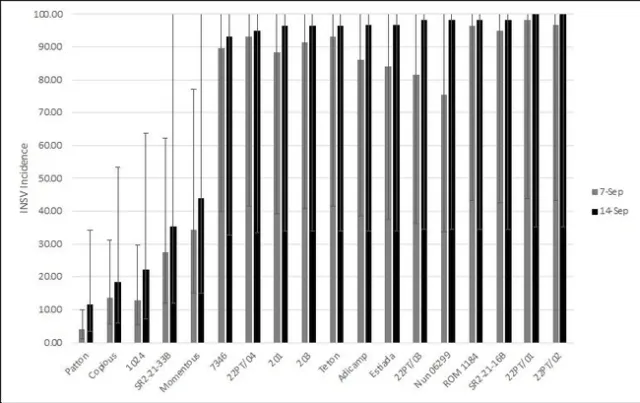
Figure 6. Mean INSV incidence (percent infected plants) of romaine lettuce on two rating dates. (Error bars represent 95% confidence intervals)
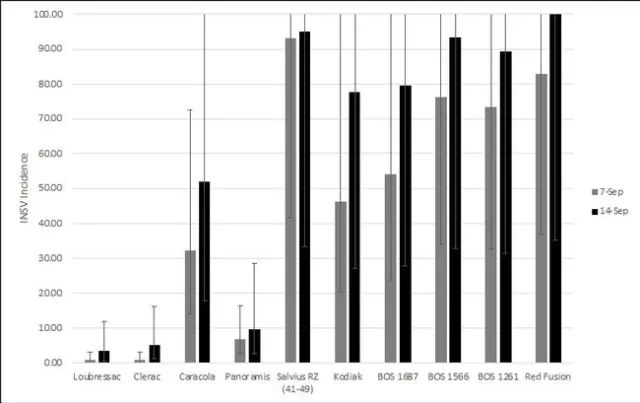
Figure 7. Mean INSV incidence (percent infected plants) of other lettuce types on two rating dates. (Error bars represent 95% confidence intervals)
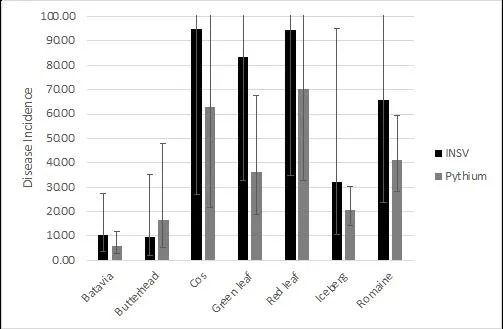
Figure 8. Mean INSV and Pythium incidence of all varieties separated by lettuce type on the final rating date. (Error bars represent 95% confidence intervals)
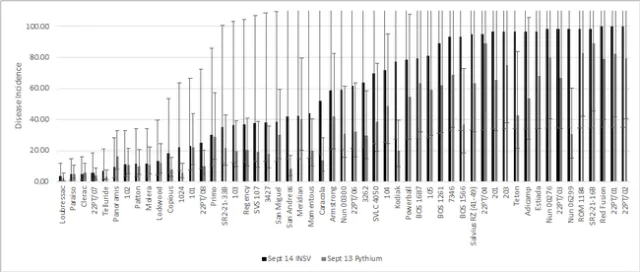
Figure 9. Mean INSV and Pythium incidence of all varieties on the final rating date. (Error bars represent 95% confidence intervals)
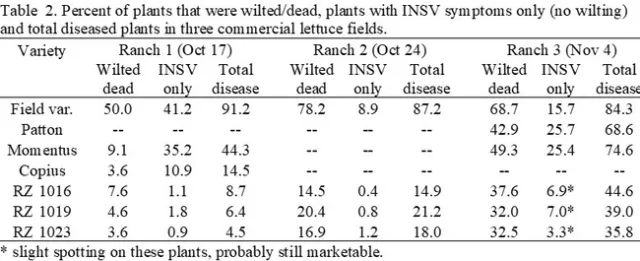
Tolerance of lettuce varieties to Fusarium wilt – 2024

Tolerance of lettuce varieties to Fusarium wilt – 2024
Alex Putman, Yu-Chen Wang, and Chris Greer
Summary
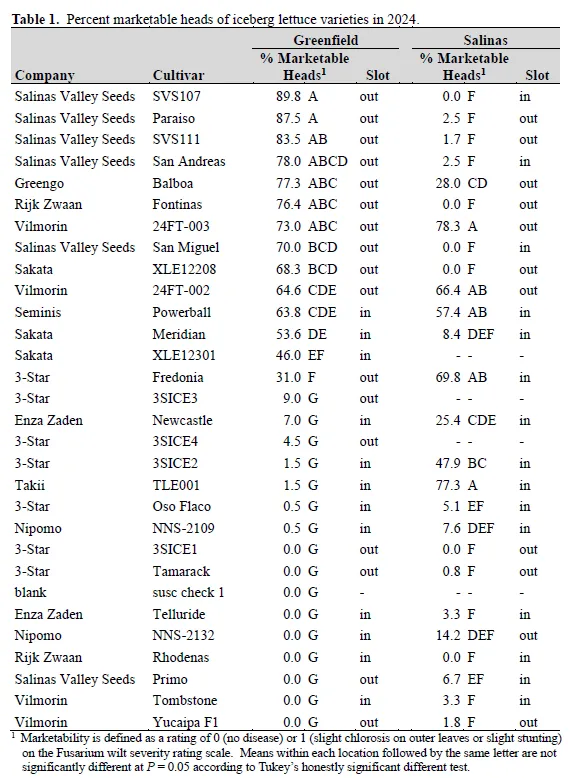
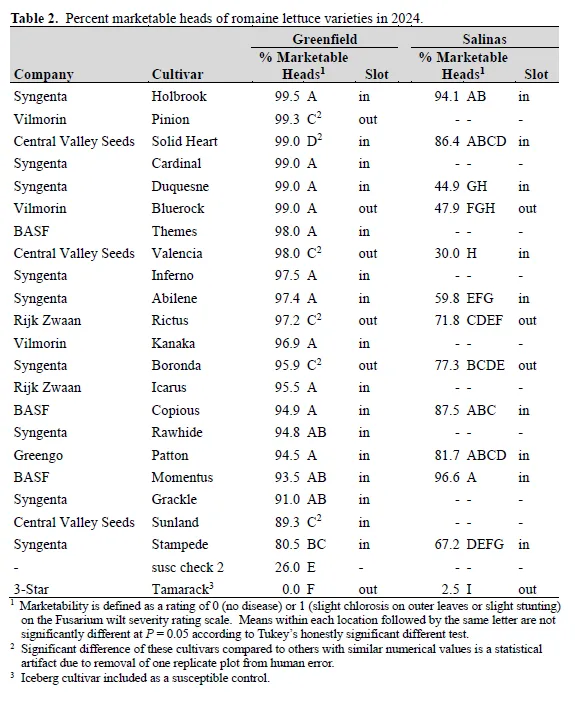
Fusarium wilt of lettuce, caused by Fusarium oxysporum f. sp. lactucae (FOL), is an economically significant disease on the Central Coast of California. We conducted field trials to evaluate 30 iceberg and 21 romaine varieties for tolerance to Fusarium wilt. The trials were located in commercial fields in Greenfield, CA (wet date of May 27, 2024) and Salinas, CA (wet date of June 8, 2024).Foliar disease severity was visually evaluated on July 31-August 2, 2024 at Greenfield and on August 5-7, 2024 at Salinas and converted to a marketability (yes or no) rating. Disease pressure was high at both locations.Iceberg varieties that exceeded 50% marketability at both locations were Powerball and two coded entries from Vilmorin-Mikado. Nine varieties exceeded 50% marketability at Greenfield but not Salinas, and two varieties exceeded the same threshold at Salinas but not Greenfield. For romaine, 19 out of 22 varieties exceeded 90% marketability at Greenfield, but only two of those varieties (Holbrook and Momentus) also exceeded the same threshold at Salinas. In a greenhouse experiment, isolates from Greenfield showed a susceptible reaction on variety Costa Rica #4, which is consistent with the Costa Rica FOL race variant. Although FOL race 1 is suspected to be present at the Salinas location, greenhouse testing is not complete. These trials provide public data on the tolerance of iceberg and romaine varieties to Fusarium wilt.
Methods
Field trials were conducted in Greenfield, CA and Salinas, CA to evaluate both in-slot and out-of-slot varieties (30 iceberg and 21 romaine) for tolerance to Fusarium wilt in commercial fields with disease history. At Greenfield, bed center spacing was 80 inches, and plots were 1 plant line wide by 100 ft. long. Due to space constraints, 4 iceberg and 10 romaine varieties were not included in the Salinas trial. At Salinas, bed center spacing was 40 inches, and plots were 1 plant lines wide by 40 ft. long. Iceberg and romaine varieties were evaluated separately, and plots of each type were arranged in a randomized complete block with four replications. Treatments were direct seeded using the grower-cooperators’ planters at Greenfield and using single-line push planters at Salinas. The wet dates were May 27, 2024 for Greenfield and June 8, 2024 for Salinas. The Greenfield trial was maintained to commercial standards for lettuce production, whereas the Salinas trial was not. After thinning by commercial crews, 50 plants at Greenfield and 30 plants at Salinas in the center of each plot were counted, and the section was marked with stakes.Data were collected from this center section. Evaluations were performed on July 31-August 2 at Greenfield and August 5-7 at Salinas, which was before maturity at the Salinas trial. Foliar disease severity was assessed on a 0 to 4 scale where: 0 = healthy; 1 = wilting or chlorosis of one to three outer leaves; 2 = up to moderate stunting and wilting or chlorosis of <25% of leaf area; 3 = head is severely stunted or absent and between 25% and 75% of leaf area is wilting or chlorotic; and 4 = head is absent and >75% of leaf area is chlorotic and nearly dead, or plant is entirely dead. For analysis, foliar disease severity was converted to marketability, where: disease severity of 0 or 1 = marketable; and disease severity of 2, 3, or 4 = not marketable. Marketability data was analyzed by an analysis of variance (P < 0.05), and variety means were separated using Tukey’s honestly significant difference test.
Results – Race of the FOL pathogen present
Two races of FOL are present on the Central Coast: race 1, and a novel race variant (Nayak et al., 2024).We are using the temporary name “Costa Rica FOL variant” for the novel race variant until it is officially named following completion of the upcoming ring test, which is a collaborative experiment between researchers and seed companies. To determine the FOL race present, isolates from each location were evaluated in a race typing experiment in the greenhouse. Variety Costa Rica #4 showed a susceptible reaction to both Greenfield isolates, which supports the observation that the Costa Rica FOL race variant is present at the Greenfield location. We suspect FOL race 1 is present at the Salinas location. However, greenhouse testing of the Salinas location isolates is ongoing.
Results – Marketability
Disease pressure was high at both locations. Iceberg varieties that exceeded 50% marketability at both locations were Powerball and two coded entries from Vilmorin-Mikado (Table 1). Nine varieties exceeded 50% marketability at Greenfield only: Balboa, Fontinas, Meridian, Paraiso, San Andreas, San Miguel, two coded entries from Salinas Valley Seeds, and one coded entry from Sakata.In contrast, two varieties exceeded the same threshold at Salinas only: Fredonia and a coded entry from Takii. This pattern of some varieties showing large differences in performance between locations whereas others showed similar performance suggests that a different race is present at each location, but this has not yet been confirmed by greenhouse testing.
Of the 21 romaine varieties evaluated, two varieties exceeded 90% marketability at both locations: Holbrook and Momentus (Table 2). A total of 17 out of 21 varieties exceeded the same threshold at Greenfield but not Salinas. At the Salinas location, Holbrook and Momentus were not statistically different from four varieties (Boronda, Copious, Patton, and Solid Heart) with average percent marketability ranging from 77% to 88%.
If you have additional questions about these trials, please contact Alex Putman at 951-522-9556 or aiputman@ucr.edu.
Please Send Us Samples
We are continuing to collect samples of lettuce Fusarium wilt to determine the distribution of races and to monitor the pathogen. To support this research, please contact the person in your region. Your help would be greatly appreciated.
- Monterey, San Benito, or Santa Cruz Counties – Yu-Chen Wang (831-201-9689 or yckwang@ucanr.edu)
- San Luis Obispo, Santa Barbara, or Ventura Counties – Chris Greer (805-888-1355 or cagreer@ucanr.edu)
- Any other California county – Alex Putman (951-522-9556 or aiputman@ucr.edu)
Acknowledgements
We are grateful to D’Arrigo Brothers Co. of California and an anonymous grower for the space and maintenance of the variety field trials. We thank seed producers for providing seed for the trial. Funding for this project was made possible by a grant from the U.S. Department of Agriculture (USDA) Agricultural Marketing Service. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the USDA. Funding was also provided by the California Leafy Greens Research Program.
References
Nayak, S., K.L. Richardson, A.I. Putman, N.R. LeBlanc, F.N. Martin, N. Li, and J.D. McCreight. 2024. Detection of novel pathogenic variants of Fusarium oxysporum f. sp. lactucae in California. Plant Pathology Early View. doi:10.1111/ppa.14019
Alternaria head rot (pin rot) of broccoli and research update on fungicide evaluation

Broccoli head rot, also known as pin rot, can cause significant problems, especially in fall broccoli production in Salinas Valley. Two types of head rot are affecting broccoli, including bacterial head rot and Alternaria head rot (Koike 2010). Here we focus on the Alternaria head rot, caused by the fungi Alternaria spp.
Symptoms. All aboveground parts of broccoli are subject to infection including heads and leaves. Head rot symptoms start as yellow spots and then turn brown and black (photo 1). The infection can spread from buds to stems (photo 2). With secondary bacteria or fungi infection, further decay occurs.The initial yellow spots resemble brown bead (photo 3), a broccoli disorder that can potentially be caused by excessive temperature, poor growth, or nutrient and water deficiency. However, the brown bead doesn't rot the stem, and no sign of fungi is presented on the buds. For uncertain cases, scraping the buds to see if the stem rot or fungi are presented is a useful technique. Leaf spot symptoms start as small yellow spots on the old leaves and then form dark, concentrical rings like a target (photo 4). The old spots may become brittle and split open or fall out as shot holes. The high number of leaf spots per plant indicates a higher disease pressure and could be a signal for fungicide application.




Management. The disease is favored by prolonged wetness from rain, dew, and fog. Fungal spores are spread by winds and splashing water. Cultural practices to promote leaf drying or prevent leaf wetness may reduce disease severity. Some growers have seen the benefits of using drip irrigation instead of overhead irrigation. An early harvest before rainfall could also reduce disease risk. Variety effects on disease tolerance might play a role. Lumpy broccoli heads tend to accumulate water which may further weaken the plant tissues and become a suitable target for the pathogens. Finally, there are a number of fungicides that have activity against the disease. More frequent fungicide applications should occur during wet weather which is favored by the disease.
Research update: fungicide evaluation.
Methods. One fungicide trial was conducted in a commercial broccoli field to test the efficacy of select fungicides for controlling broccoli head rot in 2023 fall. Broccoli ‘Centennial' were direct seeded on 27 July, 2023. Seven fungicide treatments and a nontreated control were arranged in a randomized complete block design with four replications. Each plot consisted of two seedlines of broccoli that was 30-ft long on the 40-inch wide bed. On each side of the plot was a nontreated guard bed. Treatments were applied with a CO2-pressurized backpack sprayer calibrated to deliver 35 gpa at 30 psi using a double TeeJet 8004E flat fan nozzles. Fungicide applications were made on 4 October and 16 October. All treatments were applied with non-ionic surfactant Dyne-Amic 0.08% v/v. Alternaria head rot incidence was evaluated at harvest on 23 October, 2023. Disease incidence was expressed as the percentage of the number of plants with Alternaria head rot in the total number of plants within the middle 15 ft of the plot. Data were analyzed using analysis of variance (ANOVA) and the Tukey test to separate means at P<0.05. The total rainfall received one month before harvest was 0.57 inches. The average, minimum, and maximum temperatures were 62°F, 53°F, and 75°F, respectively.
Results (Table 1). The disease pressure in this trial area was low with nontreated control having 14.0% head rot. However, significant differences occurred among treatments for the % Alternaria head rot. All treatments reduced % Alternaria head rot numerically, while Pydiflumetofen+Fludioxonil, Azoxystrobin, Fluxapyroxad+Pyraclostrobin, Fluopyram+Trifloxystrobin, and Pyraclostrobin had significantly lower % Alternaria head rot than nontreated control. And they had statistically similar % Alternaria head rot. These results also showed that single FRAC 11, premixes with FRAC 7 and 11, and premixes with FRAC 7 and 12 provided good control of Alternaria head rot; single FRAC 7 provided fair control of Alternaria head rot.
Table 1. Disease incidence of Alternaria head rot at harvest
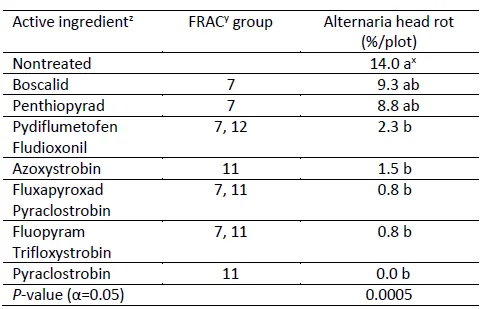
zProduct and Rate/A in this trial: Boscalid (Endura 9 oz), Penthiopyrad (Fontelis 30 fl oz), Pydiflumetofen+ Fludioxonil (Miravis Prime 11.4 oz), Azoxystrobin (Quadris 15.5 fl oz), Fluxapyroxad+ Pyraclostrobin (Priaxor 8.2 fl oz), Fluopyram+ Trifloxystrobin (Luna Sensation 7.6 fl oz), Pyraclostrobin (Cabrio 16 oz). xNumbers in a column followed by the same letter are not significantly different based on Tukey's significant difference test (P<0.05). yFRAC: Fungicide Resistance Action Committee.
Thanks to the cooperating growers and PCAs for assisting the trial. Thanks for the technical assistance from Carlos Rodriguez.
